How Prof Richard Robson won 2025 Nobel Prize in Chemistry
 Quick Read
Quick Read
The 88-year-old professor of Chemistry at University of Melbourne's discovery has opened new frontiers in sustainable chemistry.
Professor Richard Robson has won the 2025 Nobel Prize in Chemistry for helping create molecular scaffolds known as metal–organic frameworks — crystal-like structures so advanced they can pull water from desert air, trap carbon emissions, and clean our planet’s air and water.
The 88-year-old professor of Chemistry at University of Melbourne’s discovery has opened new frontiers in sustainable chemistry.
“My name is Richard Robson. I’ve been at the University of Melbourne since 1966,” he says, with the casual precision of a man who has lived most of his life in one place. The image is homely and immediate: a long career, a kitchen table, a phone call, the India Today reports.
According to the online magazine, Robson remembers the night the Nobel committee rang — “I did finish my fish. It was a bit cold, but I got there. And then I had to do the washing up,” he said — and the domestic detail does more than amuse. It pins an extraordinary moment to the ordinary rhythms that shaped it.
Robson shares the 2025 Nobel Prize in Chemistry with Susumu Kitagawa and Omar M Yaghi “for the development of metal–organic frameworks” — a type of material built like a microscopic scaffold, with lots of empty space inside.
Imagine a building made of tiny corridors and rooms that only molecules can enter. By changing the parts used to build the scaffold, chemists can make the rooms act like traps, filters or tiny reaction chambers.
That flexibility is what makes these materials useful: they can hold gases, pull water out of dry air, catch pollutants or help speed up chemical reactions in ways that ordinary materials cannot.
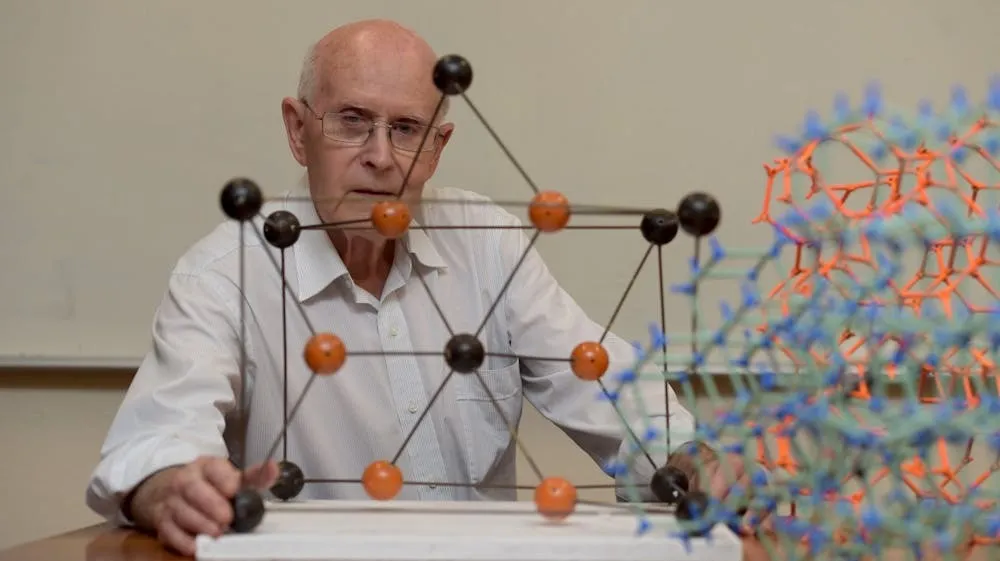
Robson tells how, in the 1970s, his teaching duties included making wooden models for first-year lectures: spheres with precisely drilled holes, connected by rods to show atomic geometry.
As he assembled those models, “the thought arose, what if we used molecules in place of the balls and chemical bonds in place of the rods? And everything else follows from that.”
The leap was simple to picture: replace the wooden balls with specially shaped molecules, and the rods with chemical links, and the same geometry would appear at a molecular scale.
The spaces inside those molecular lattices are like tiny rooms — too small to see, but big enough to hold gas molecules or small liquids. That throwaway sentence is the origin story — a leap from physical model to molecular architecture.




Comments